A CLOCK mRNA-targeting long non-coding RNA guides innate immunity training in tuberculosis
- Volume
- CitationNazimek K, Dubna Y, Bryniarski K. A CLOCK mRNA-targeting long non-coding RNA guides innate immunity training in tuberculosis. ExRNA 2026(1):0002, https://doi.org/10.55092/exrna20260002.
- DOI10.55092/exrna20260002
- CopyrightCopyright2026 by the authors. Published by ELSP.
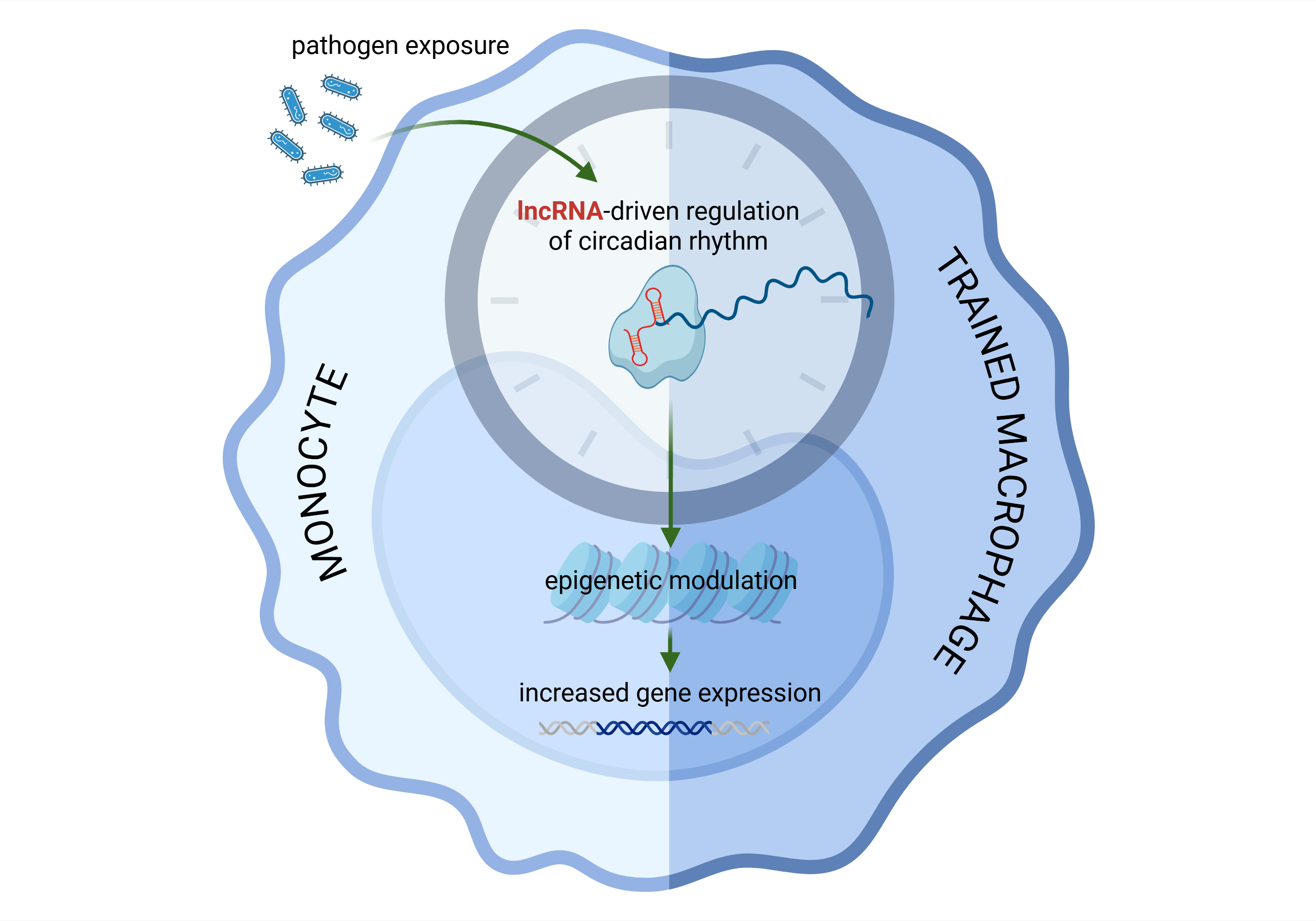
Recent years have brought the groundbreaking discovery of trained innate immunity, characterized by enhanced monocyte, macrophage and NK cell microbicidal activity. A functional shift of the involved cells results from metabolic rewiring and epigenetic modifications that, among others, are driven by long non-coding ribonucleic acid (lncRNA)-induced effects. However, many questions remain unanswered regarding the precise molecular pathways that ensure trained immunity. In this Commentary article, we aimed to present a comprehensive summary of the key findings from a recently published study that identified a role for lncRNA in monocyte training in the context of tuberculosis. Interestingly, this extracellular vesicle-transferred lncRNA has been shown to stabilize clock circadian regulator (CLOCK) mRNA and, in turn, augment its translation. Consequently, CLOCK-induced histone acetylation upregulates the expression of immune and circadian genes in trained monocyte-derived macrophages. These lncRNA-induced effects were demonstrated to increase antimicrobial resistance after immunization with the Bacillus Calmette-Guérin (BCG) vaccine as well as during unrelated infections. These latter features are desirable characteristics of trained immunity.
circadian rhythm; epigenetic regulation; extracellular vesicles; lncRNAs; monocytes; macrophages; trained innate immunity; tuberculosis

 X
X Facebook
Facebook LinkedIn
LinkedIn Reddit
Reddit Bluesky
Bluesky